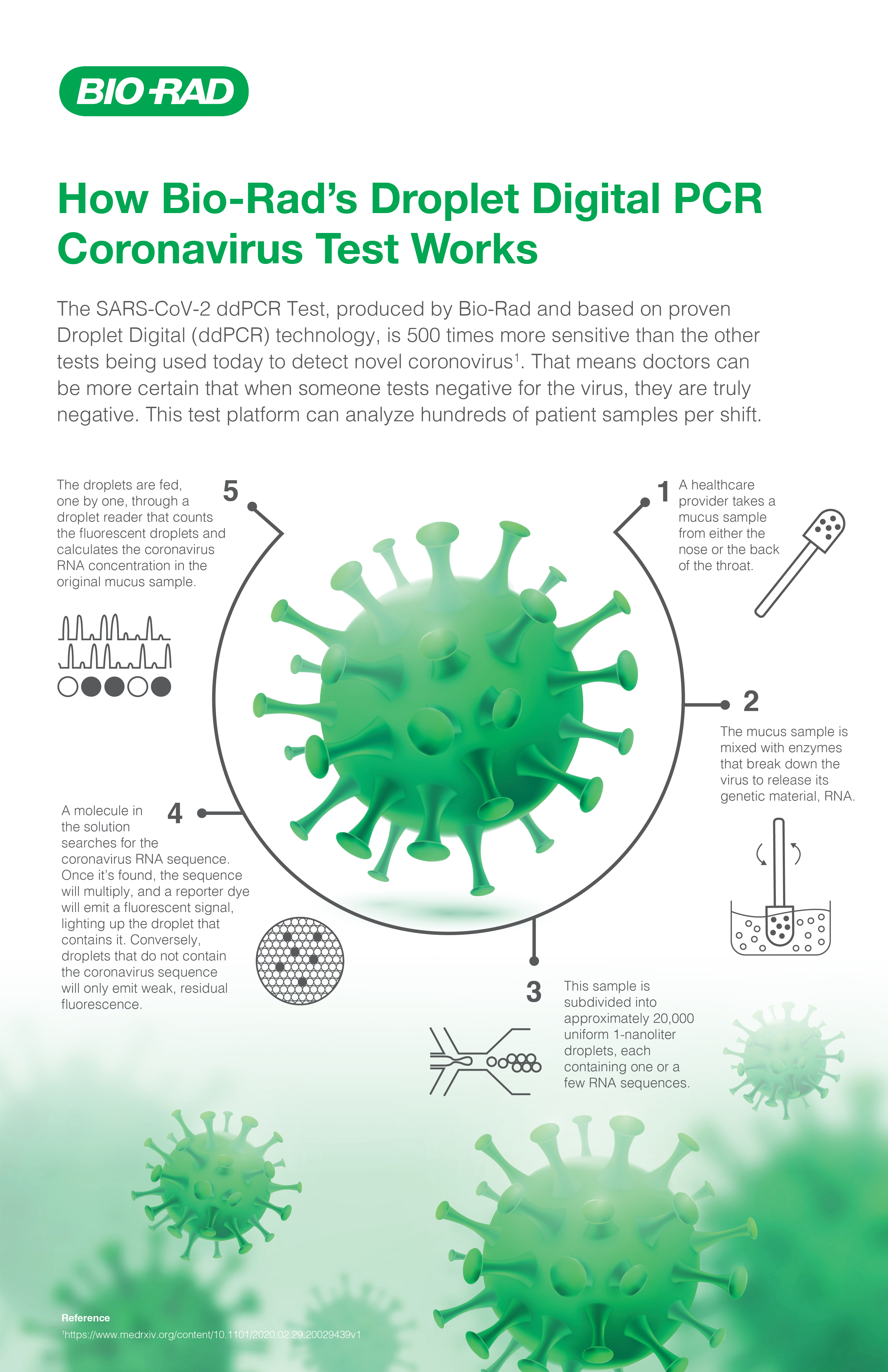
Linearity FLQ HbA1c for Bio-Rad® Systems, Calibration Verification Material, Linearity Test | AUDIT MicroControls, Inc.

NIDCD grantees to develop new smell and taste tests to screen for COVID-19 and possible future viral diseases | NIDCD

Evaluation of a rapid antigen test (Panbio™ COVID-19 Ag rapid test device) for SARS-CoV-2 detection in asymptomatic close contacts of COVID-19 patients - ScienceDirect

RadRunner review: The first test of Rad Power Bikes' new $1,299 mini-bike style electric commuter | Walk-Bike Cupertino
Bio-Rad's SARS-CoV-2 (COVID-19) Serology Test Granted FDA Emergency Use Authorization, the First Total Antibody Test to Receive EUA from the FDA - BioTechniques

Rapid Scaling Up of Covid-19 Diagnostic Testing in the United States — The NIH RADx Initiative | NEJM